-
Adding Incapacity to Injury: A Case of Trauma and Central Cord Syndrome
Christopher Bender and Jesse Powell
Introduction Central cord syndrome occurs with a lesion or soft tissue injury affecting the central anatomy of the spinal cord, causing weakness more prominent in the arms than legs, and sensory abnormalities at the level of the lesion. Here I present a case of central cord syndrome caused by a traumatic fall.
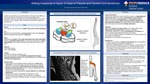
Case Patient A 71 year oldman with history of ESRD, CAD, CHF, and atrial fibrillation HPI Admitted to the hospital after a syncopalevent at home with traumatic falland subsequent development of weakness Exam Weakness was greater in the upper extremities than lower extremitiesand had a left sided predominance. This was accompanied by a severe hyperesthesiaof the upper extremities with severe pain noted on light touch Objective Vital signs: Normal Labs: Unremarkable Imaging CT head: No evidence of bleed or CVA MRI C spine (pictured at right): Multilevel degenerative changes; significant stenosis at C3-C4 with complete effacement of the thecalsac and mild cord compression Clinical Course •Neurosurgery was consulted symptoms attributed to central cord syndrome, not cervical stenosis; invasive intervention was deferred •Syncope was attributed to hypovolemia from persistent vomiting •Weakness slowly improved over the 12 days of hospitalization with physical and occupational therapy •Hospital course was complicated by urinary retention and orthostatic hypotension •Transferred to inpatient rehabilitation for additional 13 days. •At follow up with neurosurgery a few months later, the patient’s weakness had almost completely resolved, no surgical intervention was pursued
Discussion Typical Presentation •Central cord syndrome is most commonly causedby a hyperextension injury of the neckin the setting of preexisting cervical stenosis •Other causes include syringomyeliaor a slow growing tumor •Results in predominantly upper extremity weakness greater than lower extremity weakness, along with sensory abnormalities near the level of the injury, and sometimes bladder dyfunction Pathology •Affects the anterior horn gray matter or the medial aspect of the descending corticospinaltracts, resulting in weakness. •Additionally, disruption of crossing spinothalamicnerve fiberscan result in sensory abnormalities at the level of the lesion. •In our case neurological symptoms resulted from soft tissue swelling and injury after his traumatic fall •Analogous to a concussion of the spinal cord. Management •Central cord syndrome istreated conservatively with an emphasis on physical therapy and supportive management •High dose methylprednisolone can be considered in acute spinal cord injury, but its use is controversial and not currently standard of care •This patient did not receive steroids, and had a good outcome with near complete return of function •Recovery is variable, and can occur over a period of weeks to months
Takeaways • Central cord syndrome causes sensory and motor abnormalities that preferentially affect the upper extremities more than lower extremities due to the arrangement of the neural pathways •Preexisting stenosis of the cervical spinal cord is a risk factor for the development of central cord syndrome, which can occur with a traumatic hyperextension injury •Initial treatment of central cord syndrome is nonsurgical, instead relying on therapy and supportive care while allowing swelling and inflammation to dissipate. Surgical correction of preexisting abnormalities can be considered later in the course, butshould be delayed until initial healing and recovery has occurred, and is not essential to management.
-

The Peculiar Case of Purulent Pericarditis
Laura Bennetts and Meera Jain
Introduction
Purulent pericarditis is a localized purulent infection in the pericardial space •Before the era of antibiotics, purulent pericarditis was related to complications of pneumococcal pneumonia. Now more frequently associated with thoracic surgeries, immunocompromised hosts and nosocomial blood infections.1 •This case is an unusual presentation of acute methicillin-resistant staphylococcus aureus (MRSA) purulent pericarditis with a course complicated by cardiac tamponade, opiate withdrawal, and a right ventricle (RV) laceration.
Case Presentation
History of Present Illness •Patient is a 30-year-old M active intravenous drug use (IDU) with a recent history of MRSA bacteremia, endocarditis, parasternal abscess s/p I&D, and sternal osteomyelitis with inadequate treatment duration presenting to the emergency room with 3 days of worsening pleuritic chest pain and dyspnea. Physical Exam •Physical exam notable for tachycardia, low grade fever, and decreased breath sounds. No peri-sternal tenderness. Initial Diagnostic Workup •Chest radiograph withgroundglassconcerning forseptic emboli •Computerized tomography (CT) of the chest showed nodules, anteriorsternal thickening, but no evidence ofosteomyelitis. •Preliminary blood culturespending •IV vancomycin treatment was started for presumptive recurrent tricuspid valve (TV) endocarditis with septic emboli.
Hospital Course
• Patient remained tachycardic with pleuritic pain in the setting of active heroin withdrawal. • Two days after admission, preliminaryblood cultures continued to have no growth,he became hypotensive with anuptrendingleukocytosis, prompting further evaluation. • Electrocardiogram (EKG) with marked diffuse STelevations consistent with pericarditis. • Transthoracic echocardiogram remarkable for a large fibrinous pericardial effusion.
Further Complications
Pericardiocentesis wasattempted and the catheter was placed in the RV, which led toa RVrepair, subtotal phrenic to phrenic pericardiectomy and mediastinalwashout. •During the surgery frank pus was visualized in the pericardialsac. •Pericardial fluid andtissue cultures were positive for MRSA. •Hisblood cultures remained negative. Thepatient did well in thepostoperative setting and his mediastinitis was treatedwithIV vancomycin for a total of 4 weeks.
Learning Points
• Patients with purulent pericarditis generally have non-specific symptoms, so diagnosis relies on high clinical suspicion and a pericardiocentesis for pericardial fluid analysis. •The most common organism responsible for purulent pericarditis is Staphylococcus aureus.2 •Mortality rate approaches 20-30%3 making early treatment with targeted antibiotics and aggressive pericardial drainage paramount in improving patient outcomes. •Clinicians should be aware of their cognitive biases during clinical decision making, as it can be crucial for diagnosing conditions like purulent pericarditis.
-
Cryptococcoma from disseminated Cryptococcus neoformans infection in an immunocompetent patient
Megan Block, David Hotchkin, and Amy Dechet
Background Cryptococcal infection is an infection caused by encapsulated yeasts of the Cryptococcusfungi that primarily manifests in the CNS or lungs in immunocompromised hosts. Our case describes disseminated Cryptococcus neoformansinfection and cerebral cryptococcomain an immunocompetent individual.
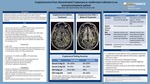
Case History •A 64 year-oldmale with no pertinent medical history presented to his PCP for worsening headaches for the past 2 months. •MRI: new ring enhancing lesion of the left thalamus •CT chest: bilateral upper lobe tree-in-bud nodules and a right upper lobe nodule with stippled calcification. •Lung biopsy, complicated by pneumothorax, was unremarkable. •2 days later developed headaches and nausea, became non-verbal, and exhibited seizure activity. Had to be intubated and sedated after this and taken to ICU. Physical Exam •Lungs: Clear. •Neuro: Does not follow commands. Opens eyes spontaneously but not to verbal stimuli. PERRL. Face appears symmetrical. Withdraws to pain in all extremities. Moves all 4 limbs spontaneously. Labs •Leukocytosis: 16.6: 88.8% neutrophils, 4.5% lymphocytes, •HIV negative •CSF: WBC 107, glucose 23, protein 79, opening pressure 23 •Cryptococcal neoformans Ag, serum EIA positive •Cryptococcal neoformans Ag, CSF EIA positive •CSF culture: Cryptococcus neoformans Management •Started on amphotericin B and flucytosine. •1 week after treatment repeat MRI brain showed improvement of the lesion, presumed to be cryptoccoma. •Required multiple LPs and drain placement due to increased ICP causing agitation and altered mental status that improved with CSF drainage. •Patient improved and discharged on 18 months of fluconazole.
Conclusions C. neoformans should still be in the considered as potential diagnosis for patient with altered mental status, regardless of immune status. •Patients can have normal radiological findings. •Consider early serum CrAg testing to expedite diagnosis. •Cryptococcomas are rare. •Treatment duration can be up to 20 months, especially for cryptococcomas. •Monitoring and reducing intracranial pressures may be key components of care.
-
A Case of Vaccine-Preventable Back Pain
Gerald Hutfles and Tricia James
The Case 55-year-old male with minimal medical history who presented multiple times over 9 months with progressive and severe gluteal & back pain, ultimately found to be from anal cancer.
HPV - The Bad, Insidious Bug
What is it? •Human Papillomavirus (HPV) is a double-stranded DNA virus of the Papillomaviridaefamily. Its lifecycle is tied to epithelial tissue. Why do we care? •HPV is associated with warts (including condyloma acuminatum) and cancer •Approximately 45,000 HPV-associated cancers occur in the US each year •7,083 -anal cancer •12,143 -cervical cancer •19,975 -oropharyngeal cancer What About Anal Cancer? •Affects ~1.6/100,000 women, and 1.0/100,000 men in the United States •Rates increase in men who have sex with men (MSM) at rates of 35/100,000 of those who are HIV-negative and 70-100/100,000 in those who are HIV-positive •5-year survival is 82% if T2N0, 42% survival if T4N+.
Anal Cancer - Decreasing the Risk
Vaccination •One studied showed an incidence rate ratio for HGSIL in females vaccinated for HPV compared to an unvaccinated population was 0.516 •Likely translates to other HPV-related disease, but further studies are needed Screening •One retrospective study -39% lower mortality risk in anal SCC in areas with higher anal dysplasia screening rates1 •No consensus on who should be screened for anal cancer and what the primary modality should be. •Recommended in HIV-infected MSM •Consider screening in: •HIV+ men & women •MSM •HIV-negative women over the age of 45 with cervical HPV 16 •Pts with history of anal warts •Women with a history of high-grade cervical, vulvar or vaginal dysplasia or cancer •Immunosuppressed patients
Learning Points
1. Anal cancer may present in an atypical fashion. It is important to consider it in the differential of buttock/back pain, particularly for patients with risk factors 2. HPV is associated with cancers that are preventable through cancer screening & vaccination. 3. No standardized methodology or guidelines exist for evaluation of anal cancer, but studies are underway. 4. Certain subpopulations with risk factors that predispose them to an increased risk of anal cancer should be screened for this disease.
-

Fentanyl-Induced Wooden Chest Syndrome Masquerading as Severe Respiratory Distress Syndrome in COVID-19
Grace Judd, Rachael Starcher, and David Hotchkin
Case Presentation
History •47 yo M with no PMH presented with fever and respiratory distress •Admitted for COVID-19 pneumonia Hospital Course •Intubated for moderate acute respiratory distress syndrome (ARDS) on hospital day 11 •Fentanyl infusion started •Hypoxia improved within 36 hrsbut plateau pressures consistently >30 cm H2O despite minimizing dead space •Trial of airway pressure release ventilation worsened hypercarbia •Bronchoscopy w/o mucous plugging, airway collapse, or purulent secretions •Lung compliance worsened (Pplat=50 on 4 cc/kg) w/o change in oxygenation •Developed suspicion for wooden chest syndrome (WCS) and discontinued fentanyl Outcome •Pplatnadired at 16 within one hour after stopping fentanyl •Pt successfully extubated on VD 10
Takeaway Points
Fentanyl uniquely activates laryngeal and respiratory muscle potentially leading to a fatal syndrome known as Wooden Chest Syndrome (WCS) manifested by poor ventilation •It is likely more common than clinicians realize Implications in practice: •Maintain a high index of suspicion for alternate or overlying physiologies when treating persistent ventilatory failure in ARDS in COVID-19 Recommendation: •Discontinue fentanyl if lung compliance and ventilatory failure seem out of proportion to hypoxia
-
Hormone Wars: The Thyroid Strikes Back! A Case of Hashimoto Encephalopathy Manifesting as Diffuse Extremity Weakness and Confusion
Cody Paiva and Brinton Clark
Case Presentation
A 42-year-old female with a past medical history of schizoaffective disorder recently discontinued on multiple psychiatric medications, stable HIV, and hypothyroidism presented with a chief complaint of diffuse extremity weakness and Parkinsonism-like symptoms. History of Present Illness •4-month history of jerking leg movements bilaterally •2-week history of ”feeling paralyzed” with difficulty ambulating •Recent long-term hospitalization at a psychiatric facility as a ward of the state •Multiple changes to psychiatric medication regimen, including abrupt discontinuation of clozapine and risperidone •Abrupt onset confusion at facility and decreased level of interaction at facility prior to admission Exam •T 98.8F, BP 114/66, P 73, RR 18, SpO2 94% on room air •Neck: thyroid without nodules or enlargement •Neuro: CN II-X intact. XI: 0/5 muscle strength on shoulder shrug or head turn. Bilateral shoulder and forearm strength 1/5 bilaterally; hip and thigh flexion/extension 0/5 bilaterally; sensation intact to bilateral upper and lower extremities •Mental status: disoriented to time, place, situation. Oriented to person.
Work Up and Hospital Course
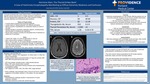
Admission Labs and Work Up: •See Table 1. Calcium 10.8, magnesium 1.6, lithium level 1.2. Other lab values unremarkable •Imaging studies without acute abnormality: CT head w/o contrast, MRI brain, MRI cervical/thoracic/lumbar spine •CSF fluid analysis: elevated protein, mildly elevated glucose •Blood, CSF, urine cultures show no growth •Continuous EEG: generalized slowing consistent with encephalopathy; no epileptiform activity •CSF negative for VDRL, HSV, VZV, EBV, CMV, fungal elements •Ativan challenge: 2mg lorazepam every 2 hours for 24 hours; if positive, suggestive of neuroleptic catatonia. In this patient, this was negative Treatment: •Dexamethasone challenge resulted in improvement of muscle weakness, confusion •Prolonged prednisone course with months-long taper
Discussion
Overview •Hashimoto encephalopathy is a rare disorder affecting patients who are commonly euthyroid, thought to be an immune complex inflammatory response in the cerebral vasculature •HE more commonly affected by women than men •It is characterized by fluctuating neurologic symptoms, most commonly behavior disturbances, confusion, and gait disturbances DIAGNOSIS •TSH can range from euthyroid to hypothyroid •TPO Ab and thyroglobulin Ab are commonly significantly elevated •CSF findings: elevated protein, lymphocytic infiltrate •EEG: non-specific slowing suggestive of encephalopathy •MRI: normal, some cases with focal hyperintensity MANAGEMENT •High dose prednisone with a prolonged course and taper; often months •>90% respond to corticosteroid therapy •IVIG and plasmapheresis have shown improvement in some cases
Conclusion
• Here we present a 42-year-old female with a rare case of Hashimoto encephalopathy manifesting as confusion and weakness •The most common clinical presentations for HE include cognitive impairment, ataxia, and focal neurologic deficits •This case demonstrates that presentation of a primary autoimmune disorder can manifest despite normal TSH and unrevealing imaging findings •This patient responded to high dose prednisone for 2 weeks with a prolonged taper and 4 days of IVIG with resolution of muscle weakness and confusion
-

Double the Battle: Two Cases of Irreversible Endocrinopathies in Patients Already Fighting Metastatic Cancer.
Olesya Petrenko and Nicholas L Stucky
INTRODUCTION: Emergence of cancer immunotherapy has instigated a new era in field of oncology and revolutionized treatment of cancer. In 2011, Immune Checkpoint Inhibitors (ICI) gained approval for treatment of advanced solid and hematological malignancies and have had remarkable results. Since being released, new cases of irreversible endocrine adverse events are increasingly becoming reported and leave patients with a new life-long diagnosis after battling metastatic cancer.
Discussion • 6 Immune Checkpoint Inhibitors (ICIs) approved: •CTLA-4 (cytotoxic T-lymphocyte-associated protein 4) inhibitor Ipilimumab •PD-1 (programmed cell death protein 1) inhibitors nivolumab, pembrolizumab •PD-L1 (programmed cell death ligand 1) inhibitors atezolizumab, avelumab, durvalumab •T-cell surface receptors immunological tolerance to self-antigens, prevent autoimmune disorders •Endocrine dysfunction –most common adverse effect •Hypothyroidism, hyperthyroidism, hypophysitis, primary adrenal insufficiency, insulin-deficient diabetes •Higher with combination therapy •Thyroid dysfunction most common •Life-threatening thyroid storm: anti-PD1 agents •Destructive thyroiditis by cytotoxic cells against thyroid gland (thyroid gland expresses both PD-L1, PD-L2 molecules) •Hypophysitismost common with ipilimumab(CTLA-4 protein expressed in pituitary glands) •Primary adrenal insufficiency –very rare (0.7%) •Insulin-dependent diabetes: with anti-PD1 therapy and Ipilimumab •Current guidelines recommend baseline screening with TFTs, morning cortisol and ACTH and screening monthly thereafter.
-

Traditional herb or drug of abuse? A case of severe Kratomwithdrawal
Nicholas Pettit, Benjamin Pedroja, and Bradford Glavan
Introduction
Kratom (Mitragynaspeciosa) is an herb native to Southeast Asia with opioid and stimulant-like properties that is commonly used to achieve a legal recreational high. •Kratom purchase and consumption is legal in most U.S. states. •Cases of acute toxicity and withdrawal have been increasing over the past decade.
Case Presentation
Pre-hospital course •Well adjusted 38 y.o. male presented with respiratory arrest requiring intubation by EMS in the field. •Found to have pinpoint pupils and bradypnea that was minimally responsive to naloxone. ICU course •Transferred to ICU and quickly extubated. •12 hours after arrival the patient became tachycardiac, tachypneic, and developed a new tremor along with new visual hallucinations. •Patient was reintubateddue to progression of symptoms. •Was restarted on his home dose of tramadol due to fear of withdrawal, symptoms persisted. •A Fentanyl drip provided minor improvement in his tremors and hallucinations.
Case resolution •On day 5, multiple empty bags of “MaengDa” tea were found in the patient’s home. •Clonidine and hydroxyzine provided further improvement in his anxiety, tremor, and tachycardia. •Extubated on day 9. •Once stable, induction therapy with buprenorphine was started and eventually discharged on day 18.
What is Kratom
History •Leaves were historically chewed by laborers in SE Asia to combat fatigue and to help treat aches and pains. Current uses •Commonly used as an opium substitute to treat pain or to treat mood disorders. •Also used to obtain a recreational high. Effects •A low doses (1-5g) has stimulant effects causing increased alertness, stamina and euphoria. •At high doses (5-15 grams) kratomacts on μ and κopioid receptors causing increase euphoria, sedation, and analgesia. •Effects begin 5-10 minsafter use and last for 2-5 hours.
Kratom Withdrawal
Withdrawal symptoms are common after discontinuing use following weeks to months of daily use. •Kratomwithdrawal begins approximately 12 hours after last use. •Duration of kratomwithdrawal is similar tothat opioid withdrawal. Symptoms of Kratomwithdrawal Treatment •There are no concrete guidelines on management of Kratomwithdrawal, however, multiple case reports have described successful treatment with symptom-triggered clonidine & scheduled hydroxyzine during acute phase. •Case reports have also described success with transitioning patients onto long term buprenorphine-naloxone maintenance.
Increase use/abuse in US
Legality of Kratom •Kratom has a long history of abuse in SE Asia and is a controlled substance in this region. •The DEA is aware of the dangers of Kratom,however it is not currently a scheduled substance. •As of 2020, there are no federal laws prohibiting Kratomuse. Purchase and consumption is legal in most states. Increasing use/abuse in U.S. •Kratomuse in the U.S. has been steadily increasing since 2010 which has caused in increase in the amountof cases of acute toxicity and withdrawal.
Learning Points
Kratom is herb with opioid and stimulant-like properties which is legal to buy and consume in most U.S. states. • Cases of acute toxicity and withdrawal from kratomare becoming increasingly common due to widespread availability in our community • Many physicians are unaware of the effects of kratom and fail to ask patients about their use of herbal medications. • Kratomwithdrawal is treatable with clonidine, hydroxyzine and buprenorphine.
-
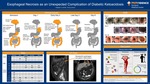
Esophageal Necrosis as an Unexpected Complication of Diabetic Ketoacidosis
Hayden Smith and Tricia James
-
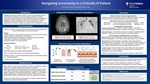
Navigating Uncertainty in a Critically Ill Patient
Brandon Tempte and Amy Dechet
Introduction Aspergillus spores are routinely inhaled.Innate immune defenses prevent fungal growth and disease in immunocompetent individuals.However, immunocompromised patients are at risk of developing invasive aspergillosis. Diagnosis of invasive aspergillus is often difficult as biopsy is not always feasible and relies on the interpretation of non-invasive testing. Early recognition and initiation of therapy is paramount as the mortality rate is high.
Case Presentation Patient is a 59 year oldfemale with a history of alcohol abuse admitted with acute encephalopathy and new onset seizure. History: -Initially presented 3 weeks earlier for jaundice, found to have acute alcoholic hepatitis, started and discharged on prednisolone -Now presenting with new onset grand mal seizure and acute confusion -Obtunded on admission and admitted to the ICU -Required intubation for airway protection Admission Workup: Chest XR: Diffuse ground glass opacities bilaterally Head CT: Patchy white matter foci in the left frontal and parietal MRI Brain (Image 1): T2/FLAIR hyperintense lesions in frontal/parietal regions in a scattered distribution TTE: EF 75% and no vegetations ALT 88 AST 140 Bilirubin 19.7
Hospital Course Day 1: Started on Vancomycin and Zosyn. TEE with no vegetations. Unable to perform LP. Unable to liberate from mechanical ventilation Day 4: Repeat MRI brain with increased size and number of lesions. Chest CT (Image 2): Multifocalair disease bilaterally with ill-defined left lower lobe density. Respiratory cultures growing aspergillus. Started on IV voriconazole Day 8: Galactomannan and beta-D-glucan positive. Repeat head CT with increased CNS lesions. Transitioned to comfort care after family discussion. Autopsy with multiple CNS fungal abscesses with leptomeningeal extension, consistent with disseminated aspergillosis
Discussion When to Test for Invasive Fungal Infections Testing for invasive fungal infection should be initiated promptly in all patients who have risk factorsand the differential includes fungal etiology. Radiographic Characteristics Pulmonary Aspergillosis:1. Single or multiple nodules (+/-cavitation) 2. Patchy/segmental consolidations 3. Peribronchial infiltrates (+/-tree-in-bud) CNS Aspergillosis:1. Ring-enhancing lesions 2. Cerebral cortical/subcortical infarction +/-hematoma 3. Sinus involvement with CNS extension How to Interpret Fungal Biomarkers and Cultures Beta-D-Glucan:Cell wall component in fungi. Found on all fugus and not specific for aspergillus. (+) test may indicate fungal infection, but not specific Galactomannan:Polysaccharide on aspergillus walls. More specific for aspergillus and can be diagnostic in the correct context. Sensitivity decreased with antifungal therapy and false (+) can occur with Zosyn. If serum results are (-), BAL can be performed, which provides additional sensitivity. Fungal Cultures:Does not always mean active infection. We are constantly inhaling conidia. (+) cultures need to be weighed again the probability of aspergillosis as the cause of disease. Biopsy is the gold standard, but often cannot be obtained in critical illness
Conclusions Invasive aspergillosis often presents with non-specific symptoms and knowing the risk factors and radiographic findings can help identify who to test -Diagnosis is based upon both identifying the organism via biopsy or biomarkers as well as probability that aspergillus is the cause of disease -Positive sputum fungal stain/culture or positive galactomannan should prompt therapy in the setting of high clinical suspicion and risk factors for aspergil
Providence Portland Internal Medicine Residency in Portland, Oregon, offers academic training in a leading community-based medical center. Our program fosters a learning environment where the intellectual rigor of an academic medical center and the cutting-edge health systems of a top community hospital merge for the betterment of both patient care and education.
Printing is not supported at the primary Gallery Thumbnail page. Please first navigate to a specific Image before printing.





